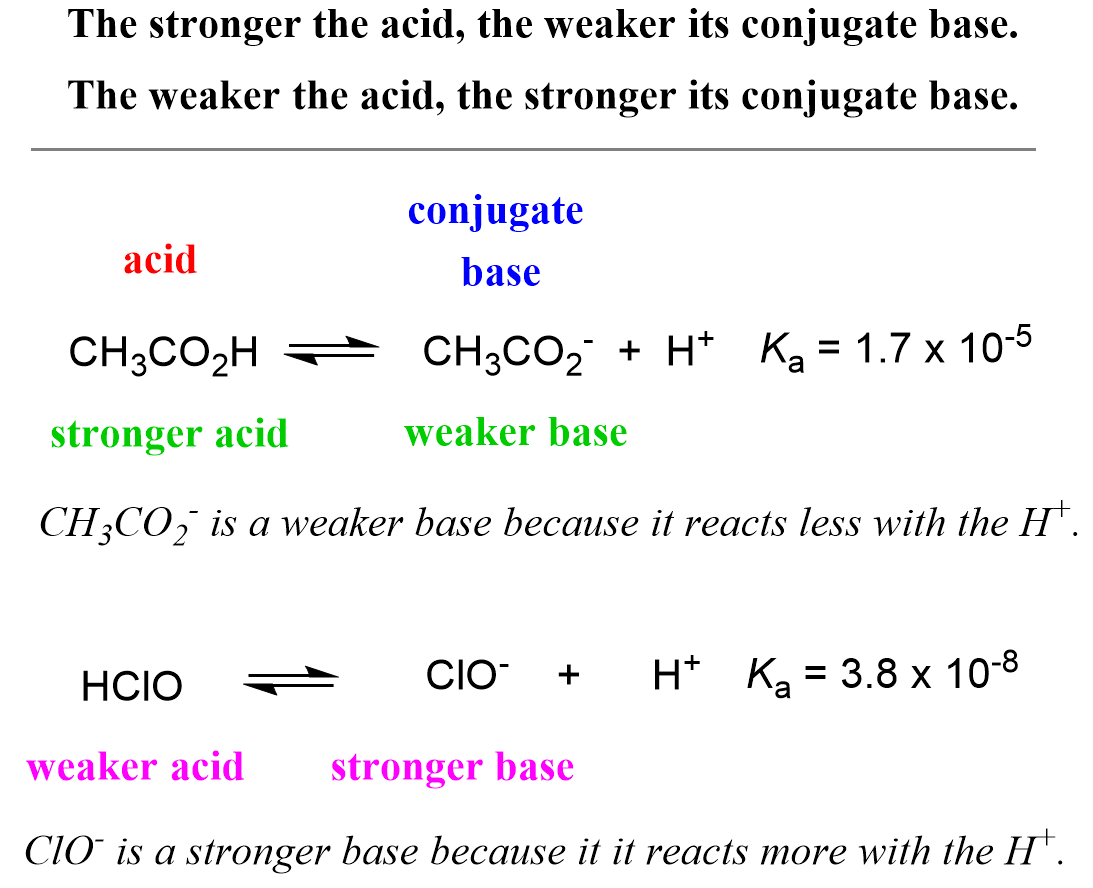
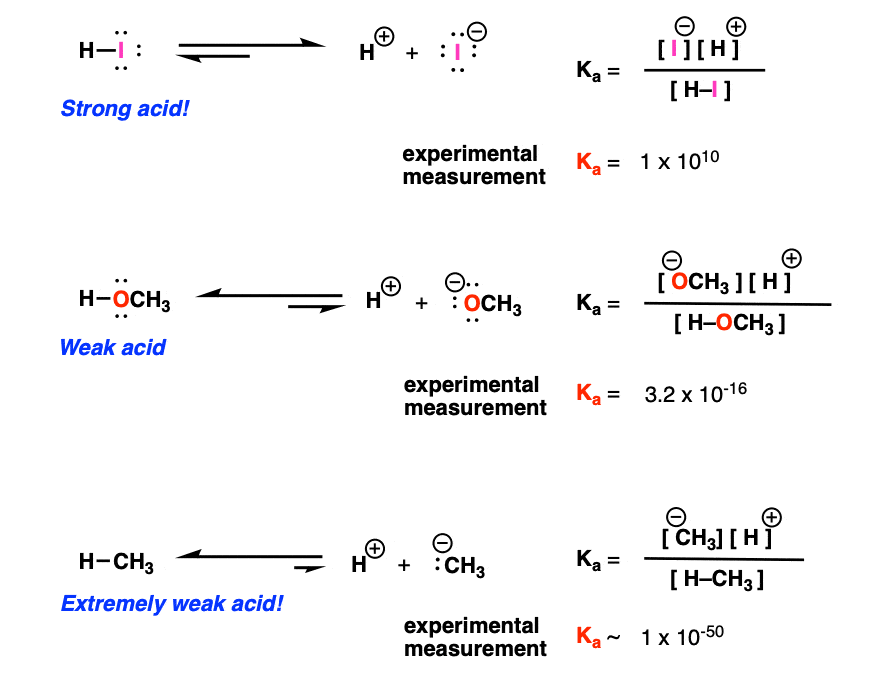Acid-Base Chemistry K a is the equilibrium constant for the dissociation of an acid into H + and its conjugate base. Ka values are typically measure in. - ppt download

Question Video: Writing an Equation for the Acid Dissociation Constant of a Generic Weak Acid | Nagwa

How To Calculate the PH of a Buffer Solution | Equation & Example - Video & Lesson Transcript | Study.com

The Relationship between K a and K b : For the ionization of the weak acid HA ; HA + H 2 O ↔ H 3 O + + A -, K
![Ka and Kb Calculations. For Weak Acid Reactions: HA + H 2 O H 3 O + + A - K a = [H 3 O + ][A - ] K a < 1 [HA] - ppt download Ka and Kb Calculations. For Weak Acid Reactions: HA + H 2 O H 3 O + + A - K a = [H 3 O + ][A - ] K a < 1 [HA] - ppt download](https://images.slideplayer.com/9/2507407/slides/slide_2.jpg)
Ka and Kb Calculations. For Weak Acid Reactions: HA + H 2 O H 3 O + + A - K a = [H 3 O + ][A - ] K a < 1 [HA] - ppt download

:max_bytes(150000):strip_icc()/what-is-pka-in-chemistry-605521_FINAL2-9fdfc39e9aa34caa96d6e74a2c687707.png)