SOLVED: A balloon has a volume of 1.80 liters at 24.0*C. The balloon is heated to 48.0*C. Calculate the new volume of the balloon a. 1.95 L b. 1.80 L c. 1.67
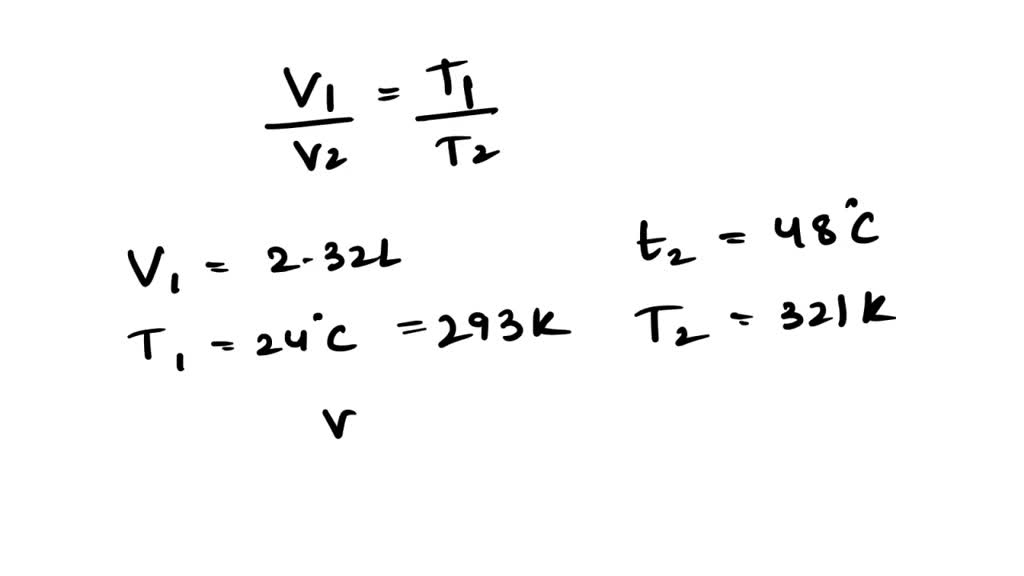
SOLVED: A balloon has a volume of 2.32 liters at 24.0°C. The balloon is heated to 48.0°C. Calculate the new volume of the balloon.

A balloon has a volume of 3.00 liters at 24.0°c. the balloon is heated to 48.0°c. calculate the new volume - Brainly.com
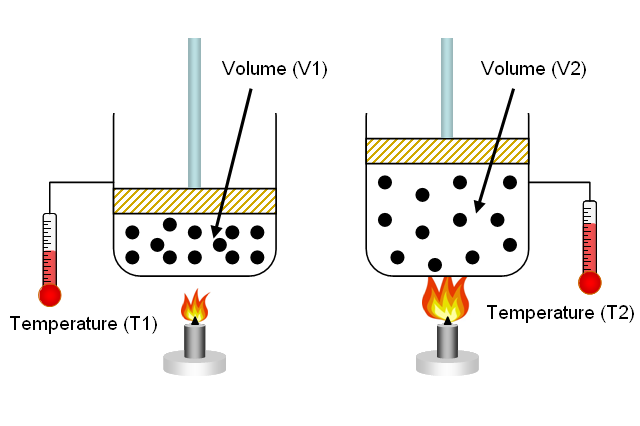
A balloon has a volume of 253.2 L at 356 K. The volume of the balloon is decreased to 165.4 L. What is the new temperature? | Socratic

Gas Law Problems #1 The temperature inside my refrigerator is about 5 ° Celsius. If I place a balloon in my fridge that initially has a temperature of. - ppt download

On heating a flask, at constant pressure, from 27% to 227^oC,200 mL of air was expelled out .what was the volume of the flask?

OpenStax College Physics Solution, Chapter 11, Problem 45 (Problems & Exercises) | OpenStax College Physics Answers

A weather balloon filled with hydrogen at 1 atm and 300 K has volume equal to 12000 liters. On ascending it reaches a place where temperature is 250 K and pressure is

Question Video: Calculating the Moles of a Gas in a Given Volume by Determining the Molar Gas Volume | Nagwa